The production of electricity can reduce the air pollution and also can compensate the global warming.
Long Description
Title:The production of electricity can reduce the air pollution and also can compensate the global warming
i am vaishali from Haryana, India here for putting forward my idea of the project not only for indian climatic conditions but can be used anywhere easily.
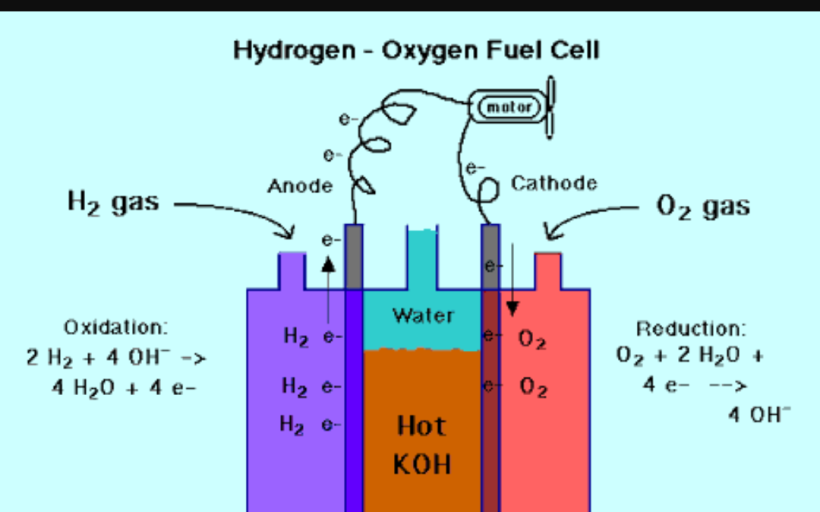
My concept and design is based on FUEL CELL ,in which we use Hydrogen gas as a fuel and Oxygen gas as an oxidant to produce the electricity . In order to the challenge the contribution for either to adapt or stop the climatic changes, I made a protocol that can contribute to three major sectors of our environment which are- producing electricity, reduction of air pollutants and the major one is Global warming.
I will not be opening all the elements of my project but I would like to explain my design a little bit further.
As in a fuel cell we use hydrogen gas as fuel and oxygen gas as an oxidant, platinum at anode as catalyst and at cathode oxygen is released and the electrolyte is also used and we get product water as well as electrical energy. The reaction goes like
At anode: A ——-> A + + e –
At cathode: A+ + B + e- ——–> AB
At anode: 2H2———>4H+ + 4 e-
At cathode O2 + 4e- + 4H+ ——–>2 H2O
This cell gives electricity continuosly and emit water as the by product.
Now about my design ….the matter is ,I have taken the structure as same as the fuel cell. I have used a gas let it be ‘A’ as fuel abd, this gas is specifically that which can be obtained from environment and also pollutes the environment. At anode the element is used whose reduction potential is more than that of gas A, so it can easily donate the e- to the anode. At cathode we have released a gas let it be ‘ B ‘ as oxidant which also do harm to our environment and has low reduction potential. We have used an electrolyte say salt bridge between anode and cathode.
The further process goes like-:
The gas A donates electrons to the anode which move towards cathode producing electric current in opposite direction. At cathode the gas B + electrons from cathode + ions of gas A which move through electrolyte towards cathode will together make such a gas which acts as cooling agent for the environment.
* The transference of electrons produce current hence generating energy.
* The use of harmful gases A and B leads to the reduction of air pollutants.
* The product made acts as a cooling agent hence compensate global warming by lowering the temperature.
Here is only the over structure and functioning of my project. The elements, gases, electrolyte used can not be revealed here.
Thank you so much for giving me the opportunity to share my idea over here.





Share on social media.
Facebook
Twitter
LinkedIn
Mail